1 : The amount of ___________ in a body depends on the nature, temperature, and mass of the body.
2 : When two bodies at different temperatures come into contact, heat flows from ___________ body to the ___________ one.
3 : Specific heat capacity is a ___________ value for a particular substance, serving as an inherent characteristic.
4 : Sea and land breezes are caused by the ___________ difference between the land and sea.
Total Questions: 4
Correct:
- The amount of ___________ in a body depends on the nature, temperature, and mass of the body. :heat energy
- When two bodies at different temperatures come into contact, heat flows from ___________ body to the ___________ one. :hotter, colder
- Specific heat capacity is a ___________ value for a particular substance, serving as an inherent characteristic. :constant
- Sea and land breezes are caused by the ___________ difference between the land and sea. :temperature
1 : ___________ is used as a coolant in car radiators and thermal power stations.
2 : The water’s high specific heat capacity releases a ___________ amount of heat energy as it ___________.
3 : The amount of heat energy required to raise the temperature of a given mass of a substance by 1°C is called its ___________.
Total Questions: 3
Correct:
- ___________ is used as a coolant in car radiators and thermal power stations. :Water
- The water’s high specific heat capacity releases a ___________ amount of heat energy as it ___________. :large, cools
- The amount of heat energy required to raise the temperature of a given mass of a substance by 1°C is called its ___________. :specific heat capacity
CALORIMETRY
Calorimetry is the science associated with determining the changes in energy of a system by measuring the heat exchanged with the surroundings for the purpose of deriving the heat transfer associated with changes.
Typical changes related to calorimetry may involve, for example,
• heating of water to its boiling point and conversion into steam
• forming of ice from water or melting of ice into water
• heating of a metal to its molten state
heat is the energy that 'spontaneously' passes between a "system", comprising, for example, matter in the form of solid or liquid, and its surroundings in some way other than through work or the transfer of matter. When a suitable physical 'pathway' exists, heat begins to flow from a 'hotter' to a 'colder' body or object.
Units of heat
Heat being a form of energy, it can be measured in appropriate units.
In CGS system, heat is measured in calories. The unit calorie is defined as "the heat energy required to increase the temperature of 1 gm of pure water through one degree Celsius at the normal atmospheric pressure" (for example, from 14.5 degrees Celsius to 15.5 degrees Celsius at STP).
one calorie = 4.184 joule.
In practice, this may be rounded to 1 calorie = 4.2 J.
A
larger unit called kilocalorie is often in use where 1 kilocalorie =
1,000 calories. One kilocalorie is defined as "the quantity of heat
energy required to change the temperature of one kilogram of (pure)
water by one degree Celsius".
British Thermal Unit (BTU}
A unit of heat energy often used,
mainly in England, is known as BTU (British Thermal Unit), defined as
the "amount of heat required to raise the temperature of one pound of
water from 63 degrees Fahrenheit to 64 degrees Fahrenheit". One BTU
equals 252 calorie (approx.).
What is Temperature?
An
exact definition of the concept "temperature" in physics is difficult
to obtain. However, an understanding of the entity called temperature
can be had in terms of The degree of 'hotness' or 'coldness' of a body or environment
- A measure of the warmth or coldness of an object or substance with reference to some standard value
- A measure of the average kinetic energy of the particles in a sample of matter, expressed in terms of units or degrees designated on a standard scale
- A measure of the ability of a substance, or more generally of any physical system, to transfer heat energy to another physical system
- Any of various standardised numerical measures of this ability, such as the Kelvin, Fahrenheit, and Celsius scales.
There are three different ways or norms, called "scales", commonly used to express temperature of a body or object
A. Fahrenheit Temperature Scale
In this scale, the temperature is denoted by the unit "degrees Fahrenheit", (symbol ° F)
B. Celsius Temperature Scale
This scale is credited to its originator, Swedish astronomer Anders Celsius(1701-1744).
C. Kelvin Temperature Scale
The special feature of this scale is that it is 273.15 units cooler than it is on the Celsius scale. Thus, a
temperature
of O kelvin is equivalent to a temperature of -273.15° C. Observe that
the degree symbol is not used with this system. So, a temperature of 300
units above O kelvin is referred to as 300 kelvin and not 300 degree
kelvin.
Note that O K is also called "absolute zero", being the lowest known temperature in practice.
Heat (energy) is frequently used to 'heat up' a body
or raise its temperature from, say, T 1 to T 2 degree, the latter being
higher, so the difference of temperature is △T = T2 - T1 in a given
unit.
It is observed that the heat input to a body depends on, or proportional to, three parameters.
• mass of the body
• difference of temperature (rise) such as △T above
• a constant that depends on the material of the body, called "specific heat"
Accordingly, the heat input or the 'quantity' of heat, Q, required for raising temperature of a body of mass m by △T degree will be given by
Q = m c △ T
where c denotes the specific heat of the material of the body, discussed later.
When m is in kg and △T is in °C, the heat input is expressed in joule (J) and the unit of c is obtained as
j-1kg-1 0C. Thus, specific heat of a substance can be defined as "the amount of heat energy required to raise the temperature of unit mass of a substance by one kelvin or 1 °C".
Thermal capacity of a substance, also referred to as
heat capacity, is the "amount of heat required to change the temperature
of an object by a certain degree".
Accordingly, thermal capacity of a
given material is an "extensive" property (whose value is proportional
to the size) and would vary with the amount of the material or
substance. For example, for the same temperature rise 100 gm of iron
will have twice the thermal capacity than that of 50 gm of iron.
The unit of thermal capacity is joule per kelvin (or JK-1) or J0C-1.
Thus, if a heat input of Q J is required to raise temperature of a material or substance by △T K, its thermal or heat capacity will be
C' = Q/△T
Heat capacity is denoted by the letter C .
Specific heat capacity, also referred to as the specific heat of a material, is "the amount of heat needed to raise the temperature of unit mass of that object by one kelvin". Using the example above, 50 gm of iron will have the same specific heat as that of 100 gm.
Accordingly, specific heat capacity or (simply) specific heat of a material or substance is "the amount of heat energy required to raise the temperature of unit mass of the material through one kelvin or one degree centigrade", denoted by the letter c.
Thus,
The method of determining the specific heat capacities of liquids or a liquid and a solid by mixing known masses of the substances at different temperatures and measuring the final temperature of the mixture is known as the method of mixtures.
Principle The principle of the method of mixtures states that the heat lost by a hot body is equal to the heat gained by
the cold body when they are mixed together and attain the same
temperature and thermal equilibrium, it being assumed that NO heat is
lost by any other process to the surroundings or otherwise.
The principle is based on the law of conservation of energy; also, sometimes called the "principle of calorimetry".
Net heat lost (by hot bodies) = net heat gained (by cold bodies or bodies at lower temperature).
Thus, let a (heat-conducting) body of mass m1 having a specific heat c1 and at a temperature of T1 is brought in 'close' contact with a body of mass m2, specific heat c2 and at (a lower) temperature T 2 such that after an elapse of sometime the two bodies together attain a steady temperature of T
where T1 >T and T2 <T
and all parameters are expressed in SI units.
Then heat lost by the hot body = m1 x c1 x (T1 - T)
and heat gained by the body at lower temperature= m2 x c2 x (T - T2)
Then, in accordance with the method of mixtures
m1 x c1 x (T1 - T) = m2 x c2 x (T - T2)
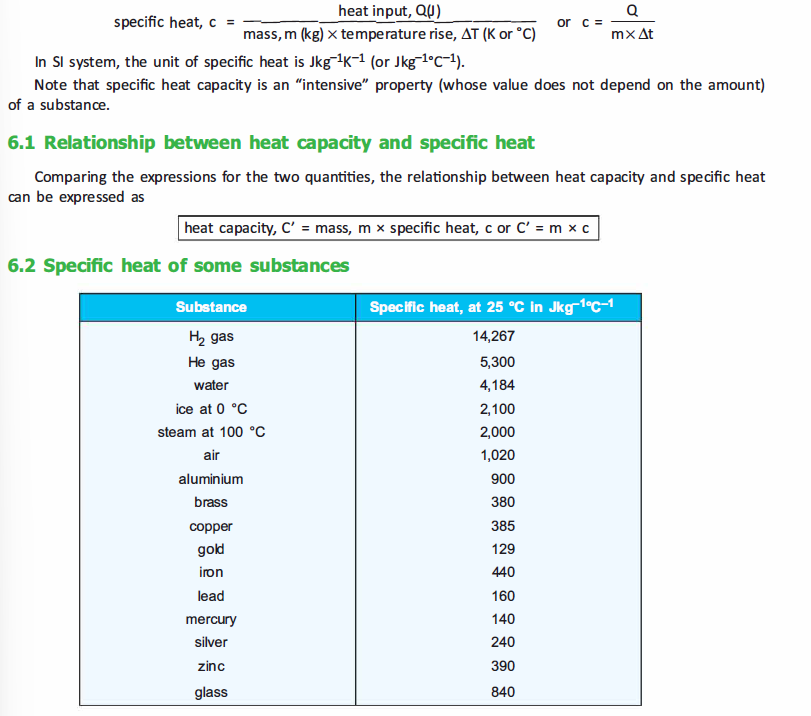
The terms melting and freezing are usually associated with the processes of change of a substance
from solid to liquid state and vice versa. The change of state from liquid to gas is termed as boiling.

Boiling occurs only when the vapor pressure of liquid and pressure of outside equals. If the pressure of
outside
increases then the boiling point of the liquid also increases. On the
contrary, if the pressure of the outside decreases, then boiling point
of the liquid also decreases. For example, at the top of a mountain the
atmospheric pressure is lower than that at the sea level and therefore
water would boil at lower than 100° C on a mountain top.
In addition to this, impurity in a liquid matter also affects the boiling point of the matter. For instance, if water is mixed with a pinch of salt or sugar, it would result in increase of boiling point of water.
So, how could there be a change in heat during a state change without a change in temperature?
During
a change in state, the heat energy is used to change the bonding
between the molecules. In the case of melting, added energy is used to
break the bonds between the molecules. In the case of freezing, energy
is subtracted as the molecules bond to one another.
These energy exchanges are not changes in kinetic energy. They are changes in bonding energy between the molecules.
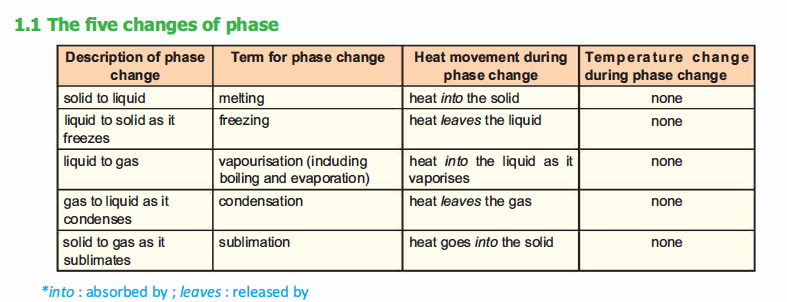
Heating curve for water
Water
has a high boiling point because of the strong hydrogen bonds between
the water molecules; it is both a strong hydrogen bond donor and
acceptor.
The heating curve for water depicting various phases is shown in Fig.2.1.
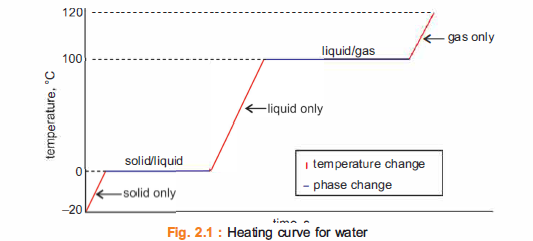
The energy is being used to loosen these attractions and change to a state of greater potential energy.
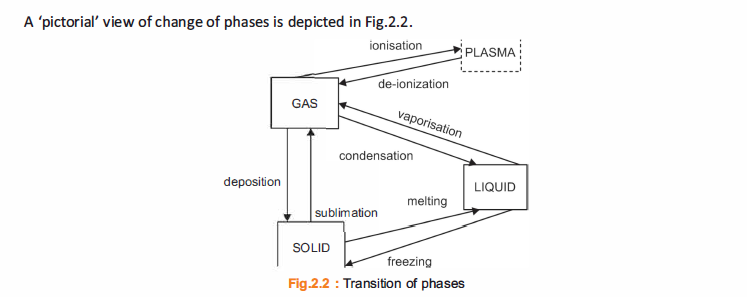
Heat absorbed or released as a result of a phase change is called latent heat, for example the heat required to convert a solid into a liquid or vapour, or a liquid into a vapour.
Accordingly, latent heat is involved in all the three cases of change of state.
Types of latent heat
There are three types of latent heat
A. The latent heat of fusion, Qf
This is the heat needed to make the change between the solid and liquid phases; for example, when
water turns to ice or ice turns to water.
B. The latent heat of vaporisation, Qv
This is the heat needed to make the change between the liquid and gas phases, as when water boils or
when steam condenses into water.
C. The latent heat of sublimation, Q5
This is the heat needed to make the change between the solid and gas phases, as when dry ice evaporates.
This is a constant for a given substance, defined as the latent heat per unit mass of the substance. The SI
unit of specific latent heat is joule per kilogram (Jkg-1).
A. Specific latent heat of fusion
Specific latent heat of fusion, Lf, of a substance is defined as the amount of heat required to change a
unit mass of the substance from solid to liquid state, without any change in the temperature.
B. Specific latent heat of vaporisation Specific latent heat of vaporisation, l,,, of a substance is defined as the amount of heat required tochange the unit mass of the substance from liquid state to gas state without a change of temperature.
The latent heat of fusion, especially that of ice, manifests itself in numerous phenomena of great natural
importance. Some of these are
■
Snow on the mountain does not melt all at once. Each kilogram of ice
would require about 336 kJ of energy to melt; this energy being supplied
from the atmosphere or the sun.
■ Cold drinks
are best cooled by adding 'soft' (not entirely crushed) pieces of ice
and not cold water at 0°C. As each gram of ice absorbs 336 J of energy
as it melts, it results in keeping the drink cold for longer.
In cold countries/climate
■ Water in the lakes and ponds does not freeze all at once as water has to absorb heat energy from the
atmosphere. The water starts to freeze first from the surface. This allows water below the surface to be
rather warm and helps fish and sea life to sustain themselves.
■ The weather becomes very cold as soon as snow starts melting as the heat is taken from the atmosphere.
■
The weather becomes pleasant when freezing of water starts as every kg
of water releases 336 kJ of heat as it turns into ice. Thus, a very
large amount of heat is slowly released in the atmosphere.■ The
temperature becomes very low after a hailstorm. This is because every
kilogram of ice absorbs 336 kJ of heat to melt, bringing down the
atmospheric temperature.■ The global warming may lead to climate
changes as a result of heat unbalance owing to melting of ice in
glaciers and polar regions.
Specific heat, or rather relative values of it, gives rise to natural phenomena in and around surroundings.
An example
Production of Sea Breeze: Along a seashore, natural breeze is produced as a result of different specific heats of the sea water and the sand on the shore. The phenomenon occurs since
• the land has lower heat
capacity than the sea water (being almost one-fifth); therefore, in day
time, the temperature of the land increases faster than the sea
• the
hot air (of lower density) above the land rises whilst cooler air from
the sea flows towards the land and hence produces sea breeze.
Compared to most substances, water has a high specific heat, being nearly 4,200 Jkg-lK-1. This has numerous useful consequences in practice.
Some of these are
Fomentation
Hot water bottles are used for fomentation, as the water remains hot in the bottle for a long time in
spite of giving off large quantities of heat.
Car radiators
Water is used as coolant in car radiators. Due to its high specific heat capacity, it can absorb a large
amount of heat energy from the engine of the car, but its temperature does not rise too high.
Bottled wines
In cold countries, wine bottles are kept under water. Due to the high specific heat capacity of water, it
takes a long time for the temperature to fall to very low level, even to 0° C.
Water in a swimming pool
Due to its high specific heat, water in a swimming pool remains cool even in summer and people enjoy
staying inside the pool.
Protection of cropsFarmers generally fill their fields with water during winter season at night to protect the crops from frost. In the absence of water, if on a cold night the temperature of the surroundings falls below 0°C, then the veins of the plants shall freeze. But water sprinkled on the crops shall not allow the temperature of the veins to fall below 0° C.
Utensils used for making tea or coffee, or cooking
vegetables or rice are made of materials of low specific heat; for
example copper, aluminium or stainless steel. In addition, they are
polished at the bottom so that they get heated quicker.
• Materials of high specific heat can be used as "insulators". Wood has a high specific heat. This allows
construction of wooden houses in countries having cold climate where wooden construction would keep
the house warm in winter, not allowing the heat of hot air from within to conduct outside.
•
The handles of utensils like a sauce pan or pressure cooker at home are
made of materials which have very high specific heat. They act as
thermal insulators. The insulating capability or conductivity is
dependent on specific heat of the material.
• In an instrument like thermometer, the body may be made of a material of high specific heat (such as
glass) and the tip or the sensor element is made of material of low specific heat (such as mercury).
• Steam has a high specific heat (more than water) and hence can carry a lot of heat energy at high pressures to run, for example, a steam turbine used as a prime mover for an AC generator.
CALORIMETER
A calorimeter also has an insulating cover or lid at the top with holes for the stirring rod and a thermometer. The lid prevents any loss of heat from the top. A photograph of an actual calorimeter is shown in Fig.1.3(a) whilst a schematic view is given in Fig.1.3(b).
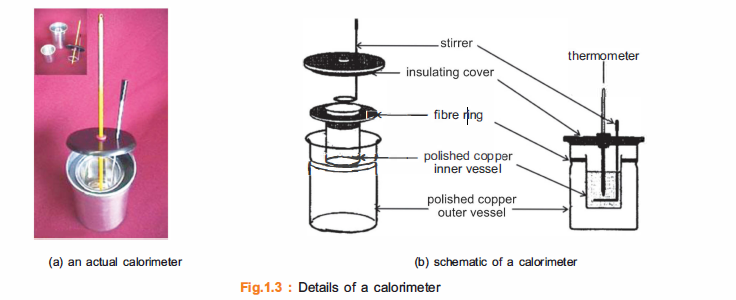
How to assemble a calorimeter
To
assemble the calorimeter, place the fiber ring in the larger vessel and
then insert the small vessel in the center of the fiber ring. Insert
the thermometer in the center hole of the lid or insulating cover;
position it so that when you close the cover, the bulb of the
thermometer will be in the center of the lower vessel, about one inch
from the bottom.
Insert the stirrer or the stirring rod and attach the handle of the insulating rod. An assembled calorimeter is shown in Fig.1.4.
An application of calorimeter
Measurement of specific heat of iron
A typical example of a calorimeter is to measure the specific heat of iron.
■
Fill the inner vessel with, say, 125 ml of water, so that its mass will
be 0.125 kg. Close the lid and leave it for about 30 minutes. At this
time the bulb of the thermometer must be in the water. (If it is not,
push the thermometer down a little). After 30 minutes, read and record the water temperature. Let it
be T1(0 C).
• Separately weigh the inner vessel of the calorimeter. Let its mass be mc kg.
• Fill up a test tube half-way with iron nails. Weigh and record the weight of the nails. Let it be mi(kg).
Place the test tube with nails in hot-boiling water for about 10 minutes. This will allow the nails to
acquire a steady temperature of 100° C without getting wet.
Open the lid of the calorimeter and transfer the nails quickly to the inner vessel. Immediately close the
lid. Move the stirrer up and down and read the temperature. Record the highest temperature. This will
be the final temperature of the water and nails. Let it be T °C.





